Fixation Challenges of Compliant Balloons – and Salt Medical’s Innovative Solution
Introduction
In modern medical device manufacturing, latex compliant balloons play a critical role across interventional catheters, dilation systems, and minimally invasive surgical tools. Their flexibility, biocompatibility, and controlled expansion capabilities make them indispensable components in many high-risk clinical applications.
However, the performance and safety of a compliant balloon depend not only on the balloon material itself, but also on how securely and reliably it is fixed to adjacent components, such as catheters, metal tubes, or connectors.
Inadequate fixation can lead to balloon detachment, rupture, or functional failure, creating serious and unpredictable clinical risks. This article explores the core technical challenges of compliant balloon fixation and introduces Salt Medical’s innovative engineering approach to overcoming them.
Industry Pain Points in Compliant Balloon Fixation
Bonding and fixing latex compliant balloons present five major industry-wide challenges:
- Material Aging
Latex is prone to degradation over time, affecting long-term bonding stability. - Stress Fatigue from High Elasticity
Repeated expansion and contraction introduce cyclic stresses that weaken adhesive interfaces. - Strict Biocompatibility & Chemical Compatibility Requirements
Any bonding method must meet stringent medical safety standards. - Complex and Sensitive Process Control
Small deviations in process parameters can lead to inconsistent bonding quality. - Difficulty Balancing Strength and Flexibility
Strong bonding often compromises elasticity, while flexibility can reduce fixation strength.
Limitations of Traditional Solutions
Conventional fixation methods, such as plasma treatment, primer application, and halogenation, each have notable drawbacks:
- Limited long-term effectiveness
- High equipment and process costs
- Safety and environmental concerns
- Difficulty meeting demanding clinical use cases
These limitations make traditional approaches insufficient for high-reliability medical applications.
Salt Medical’s Innovative Fixation Strategy
Rather than relying on additional materials or complex surface treatments, Salt Medical re-engineered the fixation concept itself.
Key Engineering Innovations
- Structural Optimization Without Adding Components
By making subtle structural adjustments, Salt Medical avoids direct bonding between latex and rigid components. Instead, fixation is redesigned as:- Soft-to-soft bonding or
- Hard-to-hard bonding
This significantly reduces stress concentration at the interface.
- Elastic Energy-Based Radial Locking
Leveraging the natural elasticity of latex, the balloon is bonded in a controlled, pre-treated state and then released, creating a radial locking force that provides dual fixation security. - Self-Reinforcing Structural Design
Internal tensile stress generated during balloon stretching is converted into a locking force, meaning: The more the balloon is pulled, the tighter it locks.
This design dramatically improves self-stability, durability, and safety.
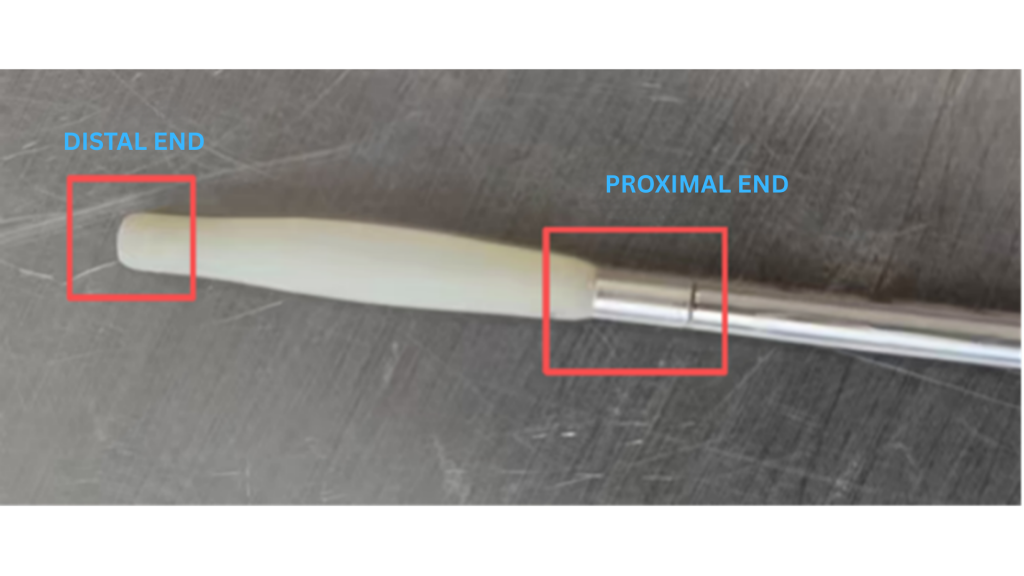
Case Study: Improving Fixation of a Compliant Dilation Balloon
To validate the effectiveness of this approach, Salt Medical applied the solution to a compliant balloon used for soft-tissue dilation and retraction.
Application Requirements
- Balloon must be fixed at both proximal and distal ends
- Ends are connected to metal tubular components
- Must withstand significant tensile forces during use

To protect the intellectual property rights of our clients, the illustrations in the case study have been desensitized. Please contact the Salt Medical team for customized solutions.

To protect the intellectual property rights of our clients, the illustrations in the case study have been desensitized. Please contact the Salt Medical team for customized solutions.
By using the improved structure proposed by Salt Medical, the bonding strength at both the proximal and distal ends significantly increased and met usage requirements:
- No additional components were added to the structure, ensuring strong compatibility.
- No special processes were required in the manufacturing, facilitating easy implementation.
- Performance-wise, bonding strength and durability were greatly enhanced, meeting the demands of high-risk scenarios.
- Cost-wise, the total lifecycle cost decreased by 15%-20% through reduced equipment investment and defect rates.
Partner with Salt Medical
Compliant balloon fixation is just one of many complex challenges in advanced medical device manufacturing. Salt Medical combines engineering expertise, process innovation, and a global development footprint to help partners move medical technologies from concept to clinical reality, faster and safer.
For more insights or customized balloon process solutions, contact Salt Medical today.
About Salt Medical
Salt Medical is your trusted medical innovation partner, accelerating the translation of cutting-edge medical technologies into clinical applications through professional engineering solutions and global collaboration.